It is a device used for colorimetry. It measures the absorbance of different wave lengths of light in a solution. Also it is used to measure the concentration of a known solute. We know that different chemical substance absorb different wave lengths of light. According to Beer-Lambert law, when the concentration of the solute is more, it soaks up further light in a specific wavelength. The simple Colorimeter Diagram is given below.
The most important parts of a calorimeter are,
a. A light source (Filament lamp)
b. An aperture which can be adjusted.
c. A set of filters in different colors.
d. A detector which measures the light, detector is usually a photo-resistor.
e. A meter which is used to display the output from detector.
In modern colorimeters the filament, light and filters may be changed by several different colored LEDs.
a. Filters: Different filters (variable optics filters) are utilized in the calorimeter to select the wave length of light which the solute takes up the most. The usual wave length sort is from 400 to 700 nanometers. Solutions are usually placed in glass or plastic cuvettes. In a manual calorimeter the cuvettes are put in and separated by hand.
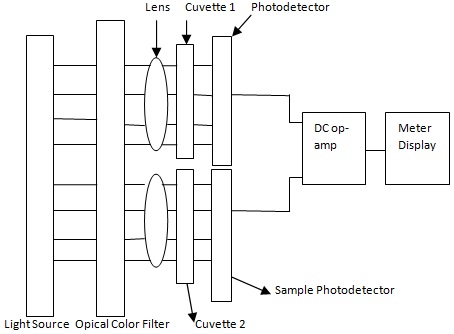
The detailed block diagram representation of a colorimeter system is given below in figure.
b. Output: The output of a colorimeter might be shown in chart or board by an analog or digital meter. The statistics may be printed on paper or stock up in a computer. It also demonstrates the quantity of light which is wrapped up by the solution or the quantity of light which has pass through the solution.
As told the results of colorimeter are displayed in percentage of optical filter transmittance or absorbance. The basic colorimeter analysis engross the peruse measurement of light intensity.
Transmittance is defined as,
The most important parts of a calorimeter are,
a. A light source (Filament lamp)
b. An aperture which can be adjusted.
c. A set of filters in different colors.
d. A detector which measures the light, detector is usually a photo-resistor.
e. A meter which is used to display the output from detector.
In modern colorimeters the filament, light and filters may be changed by several different colored LEDs.
a. Filters: Different filters (variable optics filters) are utilized in the calorimeter to select the wave length of light which the solute takes up the most. The usual wave length sort is from 400 to 700 nanometers. Solutions are usually placed in glass or plastic cuvettes. In a manual calorimeter the cuvettes are put in and separated by hand.
The detailed block diagram representation of a colorimeter system is given below in figure.
b. Output: The output of a colorimeter might be shown in chart or board by an analog or digital meter. The statistics may be printed on paper or stock up in a computer. It also demonstrates the quantity of light which is wrapped up by the solution or the quantity of light which has pass through the solution.
As told the results of colorimeter are displayed in percentage of optical filter transmittance or absorbance. The basic colorimeter analysis engross the peruse measurement of light intensity.
Transmittance is defined as,
T = I1/I0 X 100 %
I0 is the initial light intensity.
I1 is the first attenuated light intensity.
Also absorbance or optical density is defined as
A = log I0/I1 = log 1/T.
According to Beer’s Law the concentration of a solute is proportional to the absorbance given by,
A = aCL
Where, A = Absorbance
C = Concentration of absorbing substance
a = Absorbtivity